HELLENIC ACCREDITATION SYSTEM ANNEX F112 TO THE CERTIFICATE NO
A TLANTA SOUTHERN CRESCENT ALUMNAE PANHELLENIC ASSOCIATION 2013 SORORITYBYLAWS OF THE MSU PANHELLENIC COUNCIL MOREHEAD STATE UNIVERSITY
CONSTITUTION OF THE HELLENIC SOCIETY OF BRUNEL UNIVERSITY 1
HELLENIC ACCREDITATION SYSTEM ANNEX F111 TO THE CERTIFICATE NO
HELLENIC ACCREDITATION SYSTEM ANNEX F112 TO THE CERTIFICATE NO
HELLENIC ACCREDITATION SYSTEM ANNEX F114 TO THE CERTIFICATE NO
Υπουργείο Ανάπτυξης
Hellenic Accreditation System

Annex F1/12 to the Certificate No. 477-4
SCOPE of ACCREDITATION of the Veterinary Laboratory of Chalkis- National Reference Laboratory for Salmonella and Antimicrobial Resistance of the Directorate, Veterinary Center of Athens of the Ministry of Rural Development & Food |
|
Tested materials/ products |
Types of test/ Properties to be measured
|
Applied Standards/ Techniques to be used |
|
Microbiological testing |
||
|
1.Foodstuffs and animal feeding stuffs |
1. Detection of Salmonella spp. (excluding serovars S. Typhi & S. Paratyphi) |
ISO 6579-1:2017 |
|
2. Enumeration of microorganisms by the colony count method at 30 0 C, using the pour plate technique |
ISO 4833-1: 2013 |
|
|
3. Enumeration of microorganisms by the colony count method at 30 0 C, using the surface plating technique |
ISO 4833-2:2013 |
|
|
4. Detection and enumeration of Enterobacteriaceae, by the colony count technique |
ΙSO 21528-2:2017 |
|
|
5. Detection of Campylobacter spp. |
ISO 10272-1: 2017 |
|
|
6. Enumeration of Campylobacter spp. |
ISO 10272-2: 2017 |
|
|
2. Live bivalve molluscs |
1.Enumeration of β-glucoronidase positive Escherichia coli in |
ISO 16649-3:2015 |
|
3.Animal faeces, environmental samples from the primary production stage |
1. Detection of Salmonella spp. (excluding serovars S. Typhi & S. Paratyphi) |
ISO 6579-1:2017 |
|
2. Detection of Campylobacter spp. |
ISO 10272-1: 2017 |
|
|
3. Enumeration of Campylobacter spp. |
ISO 10272-2: 2017 |
|
|
4. Salmonella spp isolates |
1.Serotyping of Salmonella spp strains |
ISO 6579-3:2014 |
|
5. Salmonella spp & E.coli isolates
|
1.Monitoring of Antimicrobial resistance of Salmonella spp & E.coli strains with the Disk Diffusion agar test to: Ampicillin, cefotaxime, ceftazidime, gentamycin, nalidixic acid, ciprofloxacin, chloramphenicol, tetracycline, trimethoprim. |
Antimicrobial Susceptibility–Agar Disk Diffusion Test according to the valid editions of CLSI:Μ2, CLSI:M100, Eucast Clinical Breakpoint Table and article of Cavaco LM and Aarestrup FM (J.Clin. Microbiol.2009.Sep;47(9):2751-2758) |
|
2. Monitoring of Antimicrobial resistance of Salmonella spp & E.coli strains with the Minimum Inhibition Concetration (MIC) test in microplate |
Antimicrobial Susceptibility– Minimum Inhibition Concetration (MIC) – Broth Microdilution method using Sensititre microplate manufactured by TREK Diagnostic Systems, according to the valid editions of guidelines: ISO 20776-1 : 2006, CLSI: VET 01S, EUCAST epidemiological cut off values, Decision 2013/652/ΕΕ.
|
|
|
6. Muscular tissues and milk
|
1. Detection of antimicrobial agents (quinolones, cephalosporins, penicillins, macrolides, tetracyclines, aminoglycosides, sulfonamides) |
In house method (ΜΔ-07) based on LMV/RMV/BIO/P TC/15.AN Version 06/17-10-2006 Guidance of Community Reference Laboratory ANSES |
|
7. Campylobacter jejuni & Campylobacter coli isolates |
1. Monitoring of Antimicrobial resistance of Campylobacter jejuni & Campylobacter coli strains with the Minimum Inhibition Concetration (MIC) test in microplate. |
Antimicrobial Susceptibility– Minimum Inhibition Concetration (MIC) – Broth Microdilution method, using Sensititre microplate manufactured by TREK Diagnostic Systems, according to the valid editions of guidelines: CLSI: VET 01-A4,EUCAST epidemiological cut off values. |
Site of assessment: Laboratory permanent premises: Dokos Chalkis, Greece.
Approved signatories: Smpiraki Aphrodite, Valkanou Eleni, Mouttotou Niki, Katsaros Demetrios.
This scope of Accreditation replaces the previous one dated 08.05.2018.
The Accreditation Certificate No.477-4, to ELOT EN ISO/IEC 17025:2005, is valid until 17.11.2020.
Athens, February 28 , 2019
Konstantinos Voutsinas
Managing Director, ESYD
Page 3
of 3
Annex F1/12 to the Certificate ESYD
No.477-4
28.02.2019
HELLENIC ACCREDITATION SYSTEM ANNEX F115 TO THE CERTIFICATE NO
HELLENIC ACCREDITATION SYSTEM ANNEX F1B2 TO THE CERTIFICATE NO
HELLENIC ACCREDITATION SYSTEM ANNEX G11 TO THE CERTIFICATE NO
Tags: accreditation system, the accreditation, certificate, hellenic, system, accreditation, annex
- HOUGHTON MIFFLIN HARCOURT STORYTOWN 2008 GRADE 4 UNIT
- NEWS RELEASE JANUARY 29 2009 FRESH START AT TDC
- GENOCIDE IN BOSNIA BEFORE THE GENOCIDE BOSNIAHERZEGOVINA IS A
- TG2541 ÁRBOL DEL CAUCHO HULE 20090401 8
- HR PRIMJENA U EUU ODREDABA O TEHNIČKIM ZAHTJEVIMA ZA
- STRUKOVNA ŠKOLA VUKOVAR V U K O V A
- NC STATE UNIVERSITY IMPREST ACCOUNT GUIDELINES SEPTEMBER 2005 NC
- BROBERG ET AL VISCOSITY AND IRON DEFICIENCY IN CYANOSIS
- A PODNOSITELJ ICA ZAHTJEVA IME PREZIME ADRESA TELEFON
- PROBLEM 1 PLATE CAPACITOR FIG1 PLATE CAPACITOR DATA TASKS
- UNIFORM CANCELLATION AFFIDAVIT (FOR MORTGAGES AND VENDORS LIENS) STATE
- INSTITUT ZA ONKOLOGIJU I RADIOLOGIJU SRBIJE POZIV ZA PODNOŠENJE
- I UP ME (IME JEDNOG RODITELJA) I PREZIME
- HOW TO REQUEST A STRONG LETTER OF RECOMMENDATION FROM
- UMOWA ZAWARTA ROKU POMIĘDZY
- 3º ESO―EJERCICIOS DE ESTADÍSTICA 1 EL NÚMERO DE HERMANOS
- LAO PEOPLE’S DEMOCRATIC REPUBLIC PEACE INDEPENDENCE DEMOCRACY UNITY PROSPERITY
- CHAPTER 12 EARTHQUAKES STANDARDS SCSH3C COLLECT ORGANIZE AND RECORD
- G RADO DE FARMACIA PRÁCTICAS TUTELADAS FACULTAD DE MEDICINA
- SIDE 2 AF 20 BORNHOLMS REGIONSKOMMUNE KUNSTRÅDET TIRSDAG DEN
- ZARZĄD POWIATU GOLENIOWSKIEGO AKTUALIZACJA PROGRAMU OCHRONY ŚRODOWISKA DLA POWIATU
- CONDITIONS OF CONTRACT FOR SERVICES NO TERMS OR CONDITIONS
- GERIATRIA PROF DR HAB TOMASZ GRODZICKI STRATEGIA ROZWOJU SYSTEMU
- 16 REGLAMENTO DE RÉGIMEN INTERIOR Y DISCIPLINARIO CAPÍTULO PRIMERO
- THE ROMANIAN BALINT ASSOCIATION HISTORY AND PRESENT THE
- EINDTERMEN PER RAAMPLANLEERDOEL IN COMPETENTIE COMMUNICEREN JUNI 2015
- ZADATAK 1 A) VODJENJE POKLOPCA 1 PRI SPAJANJU
- !DOCTYPE HTML HTML LANGENUS HEAD META CHARSETUTF8 META HTTPEQUIVXUACOMPATIBLE
- COVENANT MEDICAL CENTER EXEMPT CATEGORY DETERMINATION TOOL DIRECTIONS FOR
- NCEA LEVEL 3 GEOGRAPHY (91426) 2014 — PAGE 2
 JĘZYK POLSKI 5A 26112020 TEMAT WIERSZ O PEWNYM CZARODZIEJU
JĘZYK POLSKI 5A 26112020 TEMAT WIERSZ O PEWNYM CZARODZIEJUŽÁDOST O UVOLNĚNÍ DÍTĚTE Z VYUČOVÁNÍ A ZE ŠD
SÚHLAS SO SPRACOVANÍM OSOBNÝCH ÚDAJOV FYZICKÁ OSOBA – UCHÁDZAČ
 FICHA DE PRESENTACIÓN DE LAS TAREAS TC06 DATOS
FICHA DE PRESENTACIÓN DE LAS TAREAS TC06 DATOS 256858DOCFIAA 20160423 183400 FINANSE PODRĘCZNIK UŻYTKOWNIKA – FIAA MAJĄTEK
256858DOCFIAA 20160423 183400 FINANSE PODRĘCZNIK UŻYTKOWNIKA – FIAA MAJĄTEKSKILL LAB IDK 1 KASUS 4 PSIK FIKES UMM
MARYLAND STATE ATHLETIC COMMISSION MINUTES WEDNESDAY AUGUST 27
ECLIPSE TUTORIAL FOR CSE465 GARRETT EARDLEY START UP 1
MACBETH ACT III STUDY GUIDE NAME DATE
 S AYI TOİ021 2015 KONU BES
S AYI TOİ021 2015 KONU BES SPECIFIC DIETARY REQUIREMENTS V5 IT IS FIFE COUNCIL’S POLICY
SPECIFIC DIETARY REQUIREMENTS V5 IT IS FIFE COUNCIL’S POLICYUCHWAŁA NR ………07 RADY MIEJSKIEJ W OLECKU Z DNIA
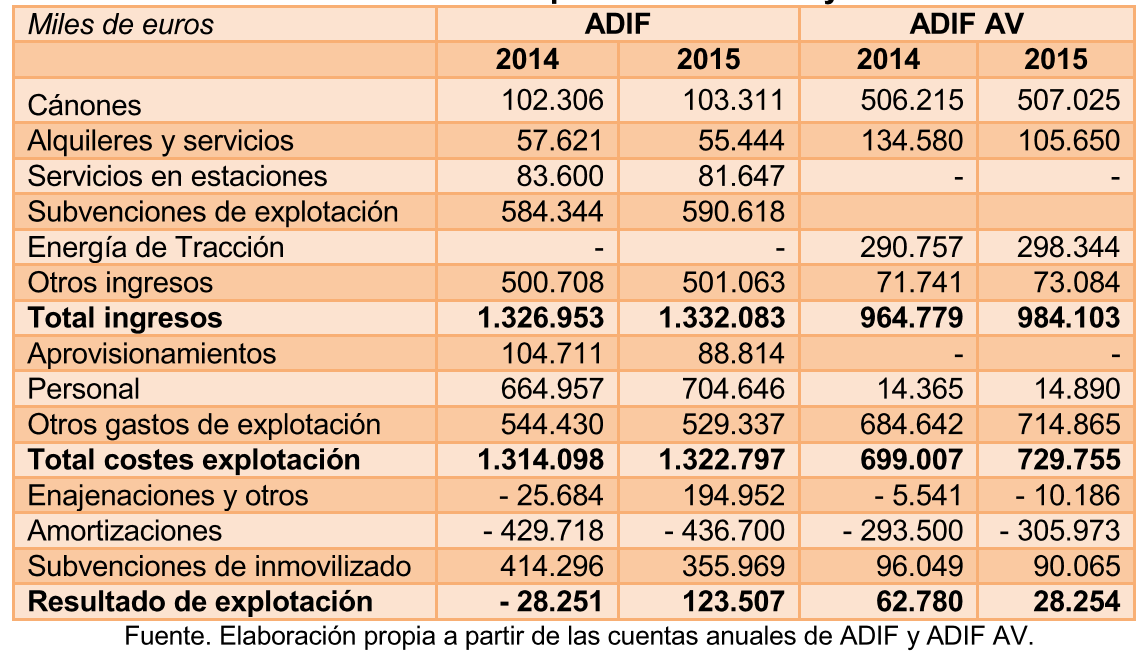 LA CNMC MODERA EL CRECIMIENTO DEL CANON PARA 2017
LA CNMC MODERA EL CRECIMIENTO DEL CANON PARA 2017 LABORATORY 6 ENUMERATION INTRODUCTION ONE OF THE CENTRAL
LABORATORY 6 ENUMERATION INTRODUCTION ONE OF THE CENTRAL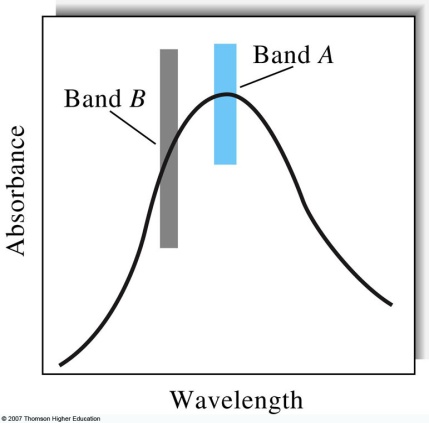 CHEMISTRY 421821 – FIRST EXAM SPRING 2009 PAGE 6
CHEMISTRY 421821 – FIRST EXAM SPRING 2009 PAGE 6 NOTA DE PRENSA 30 DE ABRIL DE 2010
NOTA DE PRENSA 30 DE ABRIL DE 2010TEDTWSIG SUBJECT RIGHT OF SOLDIERS TO PRESENT COMPLAINTS TO
INFORMACJA O MONITORINGU WIZYJNYM PROWADZONYM NA TERENIE KLUBU 1
 ISSUE 20 THIS MANUAL IS INTENDED PRIMARILY FOR THE
ISSUE 20 THIS MANUAL IS INTENDED PRIMARILY FOR THE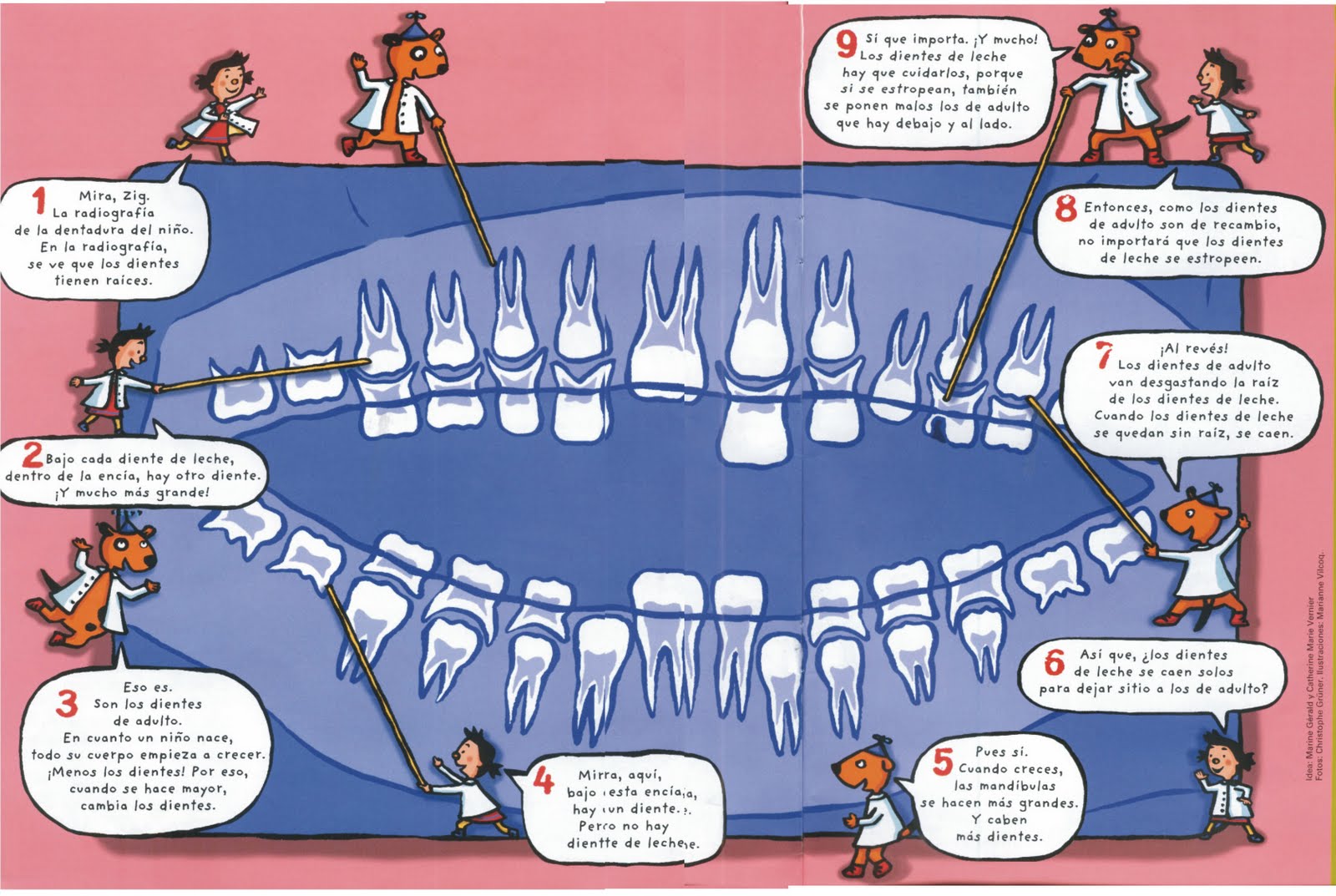 ¡¡TE INVITAMOS A NUESTRO DIA DEL NIÑO!! EL PRÓXIMO
¡¡TE INVITAMOS A NUESTRO DIA DEL NIÑO!! EL PRÓXIMO