122 COLORFUL OXIDATION OF ALCOHOL SOURCE SUMMERLIN L R
122 COLORFUL OXIDATION OF ALCOHOL SOURCE SUMMERLIN L RBE SAFE NOT SORRY CARTOON DIRECTIONS CREATE A COLORFUL
DEZEMBER 2014 FEWA COLORFUL FASHION PROJECT FEWA KOOPERIERT MIT
EAT YOUR COLORS IT’S IMPORTANT TO GET A COLORFUL
PAINT SET A SIMPLE FLUID COLORFULLY LIT SET INTERSPERSED
UNIT 4 A COLORFUL LIFE 單字測驗 1 RON
Colorful Oxidation of Alcohol
Colorful Oxidation of Alcohol
Source: Summerlin, L. R., Borgford, C. L., and Ealy, J. B. (1987)
Chemical Demonstrations: A Source Book for Teachers Volume 2. p. 210.
Description: When a yellow solution, K2CrO7, is added to three different alcohols, the solution turns blue in two of the alcohols and remains yellow in one.
Concept: Potassium dichromate will oxidize propyl alcohol and sec-butyl alcohol, but not tert-butyl alcohol. Primary and secondary alcohols are easily oxidized. The carbons holding the OH groups in propyl and sec-butyl alcohol are also holding at least one hydrogen. This hydrogen is removed by the oxidizing agent along with the H from the hydroxyl group leaving a carbonyl group and forming water.
Materials:
0.015 M K2Cr2O7 in 3 M H2SO4
propyl alcohol (1-propanol)
sec-butyl alcohol (2-butanol)
tert-butyl alcohol (2-methyl-2-propanol)
three large test tubes
25 mL graduated cylinder
Stirring rods
Safety: Wear safety goggles and gloves. Sulfuric acid is irritating. Potassium dichromate is extremely destructive to the mucous membranes of the upper respiratory tract, skin, and eyes. Exposure could be fatal. Also it is a carcinogen.
Procedure:
Add about 25 mL of each alcohol to the large test tubes.
Add 25 mL of the K2Cr2O7 solution to each tube and stir quickly.
Clean-Up: Collect the contents of the test tubes in a waste bottle, label a waste tag, and take to EH&S collection site.
Notes:
This demo has never been done in class. It was requested by Prof. Richardson for C102.
See diagram next page for information about the reactions.
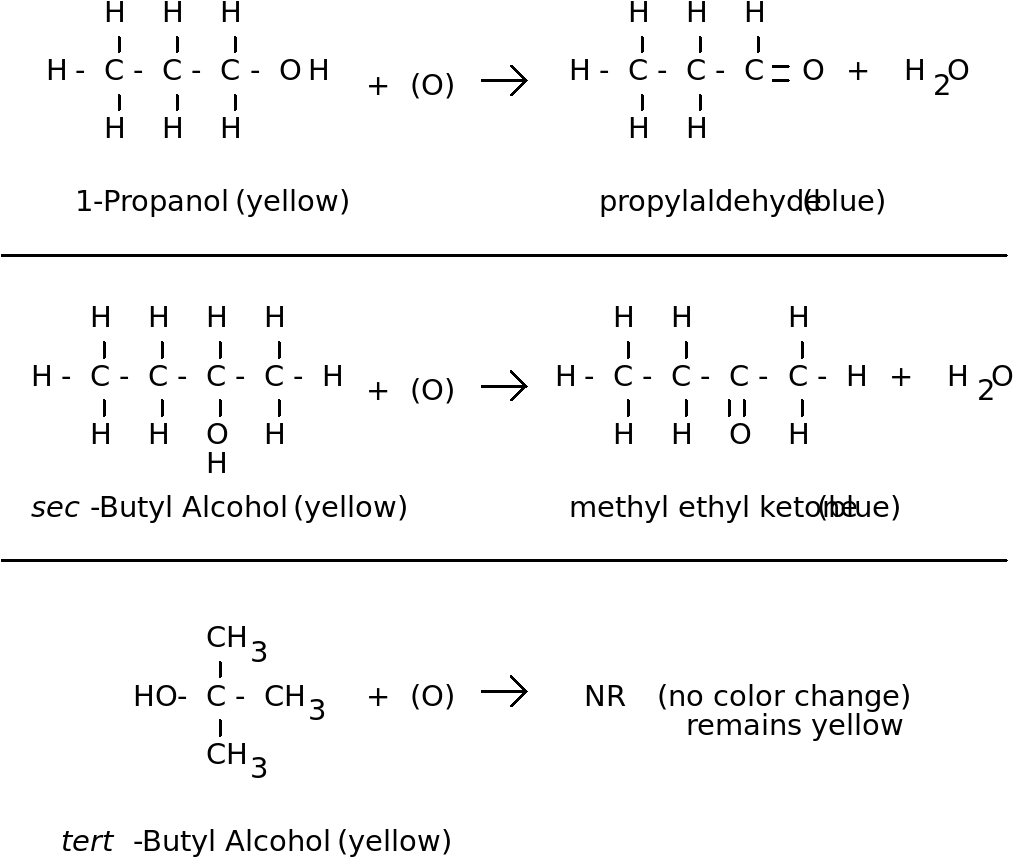
The aldehyde would be further oxidized to form a carboxylic acid. The ketone would not be further oxidized. Another demo in this manual, The Silver Mirror (Tollen's Test for Aldehydes) illustrates this principle
12-2
UNIT 4 繽紛世界 A COLORFUL LIFE READING 1 一如往常,AMBER又晚起了,且她必須跑得夠快,才能趕上黃色的校車。幸運地,她正準備過馬路時綠燈亮了,所以她趕上了。當校車經過麥當勞的金黃色拱型招
UTICA DAILY PRESS THURSDAY AUG 15 1946 COLORFUL PARADE
Tags: alcohol source:, each alcohol, source, summerlin, oxidation, colorful, alcohol
- ÅRSPLAN KLASSE 4A FAG DANSK ÅR 20142015 PERIODE HVORNÅR
- COMPARISON OF PORTABLE OXYGEN CONCENTRATORS 2012 MORE INFORMATION AND
- Ðïࡱáþÿ x81x83þÿÿÿ¥á5 ð¿ Bjbjï2ï2 `xx ÿÿÿÿÿÿx882222222fx8ax8ax8ax8aædfx¶vvvvvvvvx83wx85wx85wx85wx85wx85wx85wºxr {d©w2asvvasas©w22vv¾wwwwas 2v2vx83wwasx83wwdwkw22kwvj
- BAB II PENYUSUNAN ANGGARAN A GAMBARAN UMUM PENYUSUNAN ANGGARAN
- SERVIZO DE ALUMNADO SECCIÓN DE POSGRAO E FORMACIÓN CONTINUA
- PRIMEROS AUXILIOS Y SUPERVIVENCIA EN EL MEDIO NATURAL 1
- ATLAS FORWARD REGION FOR DISCUSSION SUGGESTED MODULE AND POWER
- RENCANA AKSI PENCAPAIAN KINERJA TAHUN 2017 SASARAN STRATEGIS 1
- UNIVERSITY OF LEEDS – CORE PSYCHIATRY TRAINING COURSE TIMETABLE
- WIPOIPGR05INF5 ANNEX PAGE 0 WIPO E WIPOIPGR05INF5 ORIGINAL ENGLISH
- EDUCACIÓN FÍSICA (SEMANA DEL 20 AL 24 DE ABRIL)
- KEMENTERIAN AGAMA INSTITUT AGAMA ISLAM NEGERI (IAIN) CURUP FAKULTAS
- BIBLIOTECA DALMACIO VELEZ SARSFIELD 03102021APASCOTTO SUPERIOR TRIBUNAL DE JUSTICIA
- GUIDELINES FOR THE ACCREDITATION OF FULL PROFESSOR PURSUANT TO
- HOJA GUIA JUGUETE BLOQUES DE MADERA NATURAL AÑO DEL
- VALGOPSTILLING TIL BESTYRELSEN I DANSK SELSKAB FOR SYGEPLEJEFORSKNING NAVN
- PROFORMA GOVERNOR’S LEARNING WALK LEARNING WALKS ARE WHERE
- LOS GASES NOBLES ISAAC ASIMOV LOS GASES NOBLES ISAAC
- PZOZZP507 OZNACZENIE SPRAWY DRUK ZP21 POWIATOWY ZESPÓŁ OPIEKI
- 1 CALCULA COMO EN EL EJEMPLO Y COMPARA LOS
- DRUŠTVO UPOKOJENIH OBRTNIKOV IN PODJETNIKOV SLOVENIJE – DUPOS PO
- NOTA DE PRENSA HOY SE PRESENTA CEGAL EN RED
- MIĘDZYNARODOWY FESTIWAL PIOSENKARZY IM HENRYKA MORYSA I ANDRZEJA LITWINA
- SEFEL LEADERSHIP SUMMIT CARAWAY DRURY INN & SUITES CONFERENCE
- SPEECH AND PRESENTATION GRADING RUBRIC PUBLIC SPEAKING AND ORAL
- INFORMACJA DLA DYSPONENTÓW ŚRODKÓW BUDŻETOWYCH NA ŚWIADCZENIA RODZINNE DYSPONENCI
- S WOPBC203 ORIGINAL INGLÉS FECHA 27 DE MAYO DE
- SUBJECT DESCRIPTION FORM SUBJECT CODE ISE2170IC2170 SUBJECT TITLE APPRECIATION
- ECONOMICS OF LOYALTY REBATES WHERE ARE WE NOW? JANUSZ
- PERSIAPAN PEMBELAJARAN DAS SALIRAWATI MSI PENDAHULUAN
DEBATE I – CUTTING CARDS ACTIVITY NAME PERIOD
 T C ÇEVRE VE ŞEHİRCİLİK BAKANLIĞI ÇEVRE YÖNETİMİ GENEL
T C ÇEVRE VE ŞEHİRCİLİK BAKANLIĞI ÇEVRE YÖNETİMİ GENEL LABORATORIO RECURSOS MEDIA PRÉSTAMO DE MATERIAL AL PERSONAL DEPARTAMENTO1
LABORATORIO RECURSOS MEDIA PRÉSTAMO DE MATERIAL AL PERSONAL DEPARTAMENTO1ESTUDIO BASICO DE SEGURIDAD Y SALUD EN LAS OBRAS
 REGIONALNA RAZVOJNA AGENCIJA POSAVJE JAVNI RAZPIS ZA IZBOR DIREKTORJA
REGIONALNA RAZVOJNA AGENCIJA POSAVJE JAVNI RAZPIS ZA IZBOR DIREKTORJAARCHIVOS CON LLAVES DE BÚSQUEDA ARCHIVOS CON ÍNDICES
 DRESSINGS ORDER FORM THIS FORM IS DESIGNED TO SUPPORT
DRESSINGS ORDER FORM THIS FORM IS DESIGNED TO SUPPORT Z NADZIEJĄ NA ZMARTWYCHWSTANIE W CHRYSTUSIE INFORMUJĘ ŻE
Z NADZIEJĄ NA ZMARTWYCHWSTANIE W CHRYSTUSIE INFORMUJĘ ŻE QUESTÕES SOBRE ALCOOLISMO PROFESSORA DA DISCIPLINA YARA TAPPIS DATA
QUESTÕES SOBRE ALCOOLISMO PROFESSORA DA DISCIPLINA YARA TAPPIS DATAWYMAGANIA EDUKACYJNE Z JEZ NIEMIECKIEGO ABY OTRZYMAĆ POSZCZEGÓLNE OCENY
 PEWIK GDYNIA SP Z OO REGULAMIN SPRZEDAŻY LIKWIDOWANYCH
PEWIK GDYNIA SP Z OO REGULAMIN SPRZEDAŻY LIKWIDOWANYCH POWERPLUSWATERMARKOBJECT357476642 TARJETA DE FIDELIZACIÓN CLUB CINEVIP A RELLENAR POR
POWERPLUSWATERMARKOBJECT357476642 TARJETA DE FIDELIZACIÓN CLUB CINEVIP A RELLENAR POR„DZIECKO I MUZYKA” PROJEKT EDUKACJI MUZYCZNEJ DLA DZIECI 56
 CHERS (ÈRES) COLLÈGUES APRÈS AVOIR TRAVAILLÉ DANS LE RÉSEAU
CHERS (ÈRES) COLLÈGUES APRÈS AVOIR TRAVAILLÉ DANS LE RÉSEAUALLEGATO 4 WESTERN SAHARA UN ENVOY OPENS THREEDAY
 ALLEGATO E DA INSERIRE NELLA “DOCUMENTAZIONE AMMINISTRATIVA” ATTO UNILATERALE
ALLEGATO E DA INSERIRE NELLA “DOCUMENTAZIONE AMMINISTRATIVA” ATTO UNILATERALEFREEDOM OF INFORMATION ACT 2000 YOUR REQUEST FOR INFORMATION
 RAINY DAYS LET’S COMPARE THE TOTAL AMOUNT OF PRECIPITATION
RAINY DAYS LET’S COMPARE THE TOTAL AMOUNT OF PRECIPITATION AMENDMENT OF AN INCORRECTLY NOTIFIED BOVINE ON FARM DEATH
AMENDMENT OF AN INCORRECTLY NOTIFIED BOVINE ON FARM DEATH7 INFORME NO 1511 PETICIÓN 22203 INADMISIBILIDAD LUIS ALBERTO